This February, the women’s health field was hit with some exciting news: pap smears may one day be a thing of the past!
I’m 22 years old, and every year I get slight shivers when realising I’m a year closer to my first cervical cancer screening appointment. As a reproductive biologist and incoming first year medical student, I’m not afraid to say that I feel anxious about it. And I know I’m not alone. Unfortunately, fear, anxiety, cultural beliefs, and negative healthcare experiences can all create barriers to cervical screening. As a result, around a third of women invited for screening in the UK do not attend. This means some cases of cervical cancer that could have been detected earlier may instead be diagnosed later, when treatment becomes more difficult.
Let’s take a breath here and backtrack. There is a lot of urgency over cervical cancer screening, but when people don’t fully understand a disease or the benefits of early detection, that urgency is easily lost in translation.
What even really is cervical cancer? Cervical cancer is primarily caused by persistent infection with high-risk types of human papillomavirus (HPV), which can cause changes in cervical cells that may eventually develop into cancer. There are many HPV types, but HPV 16 and 18 are responsible for around 70% of all cervical cancer cases (Cancer Research UK, n.d.). HPV can be transmitted through any kind of sexual activity, so practicing safe sex helps to reduce the risk transmission. Additionally, there are now vaccines that are routinely given in the UK to boys and girls aged 12/13 (Cancer Research UK, n.d.). This vaccine provides a greater chance of protection against cervical cancer, but it doesn’t protect against all types of HPV, so it is still greatly advised that people do go for the cervical cancer screenings (Cancer Research UK, n.d.).
Sometimes statistics can help bridge the gap between yourself and a disease. In the UK, cervical cancer is the 14th most common cancer in females, with 3,300 new cases of cervical cancer each year, which is around 9 every day (2017-2019) (Cancer Research UK, n.d.). Consequently, there are around 890 cervical cancer deaths in the UK every year, which is more than 2 every day (2021-2023). Fortunately, around two in three women diagnosed with cervical cancer in the UK survive for 10 years or more (Cancer Research UK, n.d.). Additionally, 99.8% of cervical cancer cases are preventable (2015). A person’s risk of developing cancer depends on many factors, including age, genetics, and exposure to risk factors (including some potentially avoidable lifestyle factors) (Cancer Research UK, n.d.).
When I think of cervical cancer screening I picture a larger-than-life, daunting, metal speculum and a woman grimacing in pain. But that’s just my brain maximising my anxieties, when in actual fact most people who have yet to go to their first screening appointment don’t know exactly what the appointment entails:

- Once undressed from the waist down, you’ll be covered by a sheet and asked to lie back on a bed with your legs bent, feet together and knees apart
- Then a speculum will gently be put into your vagina using a small amount of lubricant.
- The nurse or doctor then opens the speculum to see your cervix and will use a small soft brush to take small samples of cells.
- The nurse or doctor will then close and remove the speculum and leave you to get dressed.
The cervical screening test itself usually takes less than 5 minutes, and the whole appointment should take about 10 minutes (NHS, 2025)! I don’t know about you but that information makes things feel a lot less daunting.
A recent study from China published in the BMJ has suggested that testing menstrual blood for HPV could be a “robust alternative or replacement” for current cervical screening by a clinician (Tian et al., 2026). The study used a group of 3,068 women aged 20-54 years with regular menstrual cycles, enrolled between September 2021 and January 2025 at four urban and three rural communities in Hubei Province, China. Each participant gave three samples for testing: a menstrual blood sample collected using a minipad, a cervical cell sample collected by a clinician, and an additional clinician collected cervical sample for laboratory processing. A mobile app was also given to participants to see test results and receive advice from healthcare providers (Tian et al., 2026).
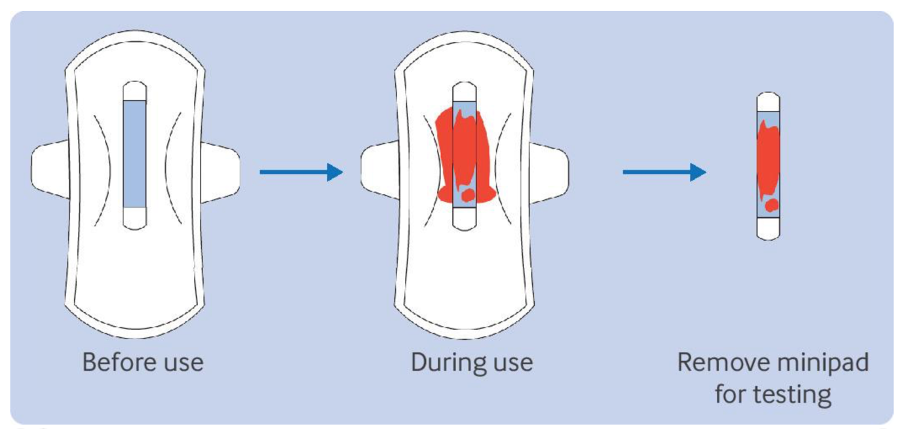
The study measured the diagnostic sensitivity and specificity. Sensitivity tells us how well a test identifies people who have a disease, while specificity shows how well a test correctly identifies people who do not. The results showed that menstrual blood samples collected using a minipad had a sensitivity of 94.7% for detecting a high-grade cervical disease, compared to 92.1% for clinician collected cervical samples (Tian et al., 2026). The minipad method had a slightly lower specificity (89.1% vs 90.0%), meaning it produced slightly more false positives. However, the probability that someone with a negative result truly did not have the disease was identical for both methods (99.9%) (Tian et al., 2026). Additionally, there wasn’t a significant difference in the probability that someone with a positive result truly had the disease between both collection methods. Although the research group acknowledged some study limitations, the results of the large-scale community-based study show promising potential for menstrual blood HPV testing (Tian et al., 2026).
The study outlined limitations of this collection method for HPV testing such as that since menstrual blood flows through the genital tract, HPV can infect sites beyond the reach of conventional sampling methods. So, while menstrual blood testing could identify more women with cervical cancer that standard cervical screening samples may miss, false positive results could also increase (Tian et al., 2026). Additionally, the accuracy of their diagnostics was somewhat limited by the study sample size, so there is a need for future studies with a greater sample size and women with higher-grade diagnoses (Tian et al., 2026).
I find this research incredibly exciting because it presents a potential alternative to standard cervical screening. A less invasive method could increase participation among women and people with a cervix who currently avoid screening due to discomfort, anxiety, or access barriers.
However, menstrual blood testing currently focuses on detecting high-risk HPV, rather than directly identifying abnormal cervical cells. Therefore, it may work best as a first step in the screening pathway, rather than completely replacing traditional cervical screening.
However, I am very keen to see where the research progresses from here and the eventual translation to medical practice.
Next week, we will be locking into a recently published collaborative study by scientists at the University of Manchester, Queen’s University (Ontario, Canada), and Cryos International (Aarhus, Denmark), showing that sperm may be happiest in the summer months – just like many of us.
REPROVA.
Cervical cancer statistics (no date) Cancerresearchuk.org. Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/cervical-cancer (Accessed: March 6, 2026).
Risks and causes of cervical cancer (no date) Cancerresearchuk.org. Available at: https://www.cancerresearchuk.org/about-cancer/cervical-cancer/risks-causes (Accessed: March 6, 2026).
Tian, X. et al. (2026) “Testing menstrual blood for human papillomavirus during cervical cancer screening in China: cross sectional population based study,” BMJ (Clinical Research Ed.), 392, p. e084831. Available at: https://doi.org/10.1136/bmj-2025-084831.
Website, N.H.S. (2025) What happens at your cervical screening appointment, nhs.uk. Available at: https://www.nhs.uk/tests-and-treatments/cervical-screening/what-happens/ (Accessed: March 6, 2026).




Leave a comment